Advances in Prostate Cancer Research
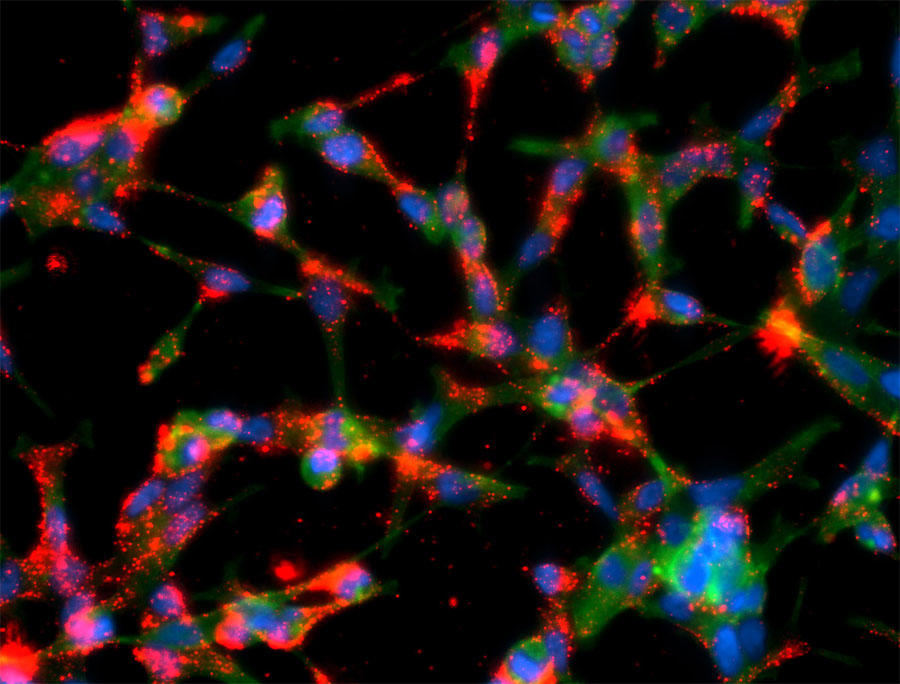
Nanoparticles are tested as a means to deliver drugs to prostate cancer cells.
NCI-funded researchers are working to advance our understanding of how to prevent, detect, and treat prostate cancer. Most men diagnosed with prostate cancer will live a long time, but challenges remain in choosing the best treatments for individuals at all stages of the disease.
This page highlights some of the latest research in prostate cancer, including clinical advances that may soon translate into improved care, NCI-supported programs that are fueling progress, and research findings from recent studies.

Studying Early Detection for Men at High Risk
Men with certain inherited genetic traits are at increased risk for developing prostate cancer. Examples of such traits include inherited BRCA gene mutations and Lynch syndrome . No clear guidelines exist for when or how—or if—to screen men at high genetic risk for prostate cancer.
NCI researchers are using magnetic resonance imaging (MRI) of the prostate in men at high risk to learn more about how often and how early these cancers occur. They’re also testing whether regular scans in such men can detect cancers early, before they spread elsewhere in the body ( metastasize ).
Diagnosing Prostate Cancer
Improving biopsies for prostate cancer.
Traditionally, prostate cancer has been diagnosed using needles inserted into the prostate gland in several places under the guidance of transrectal ultrasound (TRUS) imaging to collect samples of tissue. This approach is called systematic biopsy .
However, ultrasound does not generally show the location of cancer within the prostate. It is mainly used to make sure the biopsy needles go into the gland safely. Therefore, biopsy samples using ultrasound guidance can miss cancer altogether. Or they may identify low-grade cancer while missing areas of high-grade , potentially more aggressive cancer.
Some doctors, concerned that a systematic biopsy showing only low-grade cancer could have missed a high-grade cancer, may suggest surgery or radiation. However, in some cases these treatments will be for a cancer that may have never caused a problem, which is considered overtreatment .
Using MRI and ultrasound . Scientists at NCI have developed a procedure that combines magnetic resonance imaging (MRI) with TRUS for more accurate prostate biopsies. MRI can locate potential areas of cancer within the gland but is not practical for real-time imaging to guide a prostate biopsy. The procedure, known as MRI-targeted biopsy, uses computers to fuse an MRI image with an ultrasound image. This lets doctors use ultrasound guidance to take biopsy samples of areas of possible cancer seen on MRI.
NCI researchers have found that combining MRI-targeted biopsy with systematic biopsy can increase the detection of high-grade prostate cancers while decreasing detection of low-grade cancers that are unlikely to progress.
Testing machine learning . Researchers are testing the use of machine learning , also called artificial intelligence (AI), to better recognize suspicious areas in a prostate MRI that should be biopsied. AI is also being developed to help pathologist s who aren't prostate cancer experts accurately assess prostate cancer grade . Cancer grade is the most important factor in determining the need for treatment versus active surveillance .
Finding small amounts of prostate cancer using imaging and PSMA
NCI-supported researchers are developing new imaging techniques to improve the diagnosis of recurrent prostate cancer. A protein called prostate-specific membrane antigen (PSMA) is found in large amounts—and almost exclusively—on prostate cells. By fusing a molecule that binds to PSMA to a compound used in PET imaging, scientists have been able to see tiny deposits of prostate cancer that are too small to be detected by regular imaging.
The Food and Drug Administration (FDA) has approved two such compounds for use in PET imaging of men with prostate cancer. These approvals are for men whose cancer may have spread to other parts of the body but is still considered curable, either with surgery or other treatments.
The ability to detect very small amounts of metastatic prostate cancer could help doctors and patients make better-informed treatment decisions. For example, if metastatic cancer is found when a man is first diagnosed, he may choose an alternative to surgery because the cancer has already spread. Or doctors may be able to treat cancer recurrence—either in the prostate or metastatic disease—earlier. which may lead to better survival. Studies are being done to determine if such early detection can improve outcomes.
As part of the Cancer Moonshot℠ , NCI researchers are testing whether PSMA-PET imaging can also identify men who are at high risk of their cancer recurring. Such imaging may eventually be able to help predict who needs more aggressive treatment—such as radiation therapy in addition to surgery—after diagnosis.
Research teams are also looking at:
- whether certain patterns seen on PSMA tests taken over time may indicate an increased risk of recurrence after initial treatment.
- how small metastases discovered with PSMA change over time , with or without treatment.
New Prostate Cancer Treatments
Standard treatments for prostate cancer that has not spread elsewhere in the body are surgery or radiation therapy (RT), with or without hormone therapy .
Active surveillance is also an option for men who have a low risk of their cancer spreading. This means monitoring the cancer with regular biopsies and holding off on treatment unless there is evidence of progression. Rates of active surveillance more than doubled between 2014 and 2021 , to almost 60% of US men diagnosed with low-risk prostate cancer.
Hormone therapy for prostate cancer
Over the last decade, several new approaches to hormone therapy for advanced or metastatic prostate cancer have been approved for clinical use.
Many prostate cancers that originally respond to treatment with standard hormone therapy become resistant over time, resulting in castrate-resistant prostate cancer (CRPC). Four newer drugs have been shown to extend survival in some groups of men with CRPC. All inhibit the action of hormones that drive CRPC:
- enzalutamide (Xtandi)
- abiraterone (Zytiga)
- darolutamide (Nubeqa)
- apalutamide (Erleada)
These drugs are now also used in some people whose prostate cancer still responds to standard hormone therapies but has spread elsewhere in the body (metastasized).
Scientists are continuing to study novel treatments and drugs, along with new combinations of existing treatments, in men with metastatic and castration-resistant prostate cancer.
PARP inhibitors for prostate cancer
A PARP inhibitor is a substance that blocks an enzyme in cells called PARP. PARP helps repair DNA when it becomes damaged. Some prostate tumors have genetic defects that limit their ability to repair DNA damage. Such tumors may be sensitive to PARP inhibitors.
Two PARP inhibitors, olaparib (Lynparza) and rucaparib (Rubraca) , have been approved for some men whose prostate cancer has such genetic defects and has metastasized , and whose disease has stopped responding to standard hormone treatments. Ongoing studies are looking at combing PARP inhibitors with hormone therapies.
Immunotherapy: vaccines for prostate cancer
Immunotherapies are treatments that harness the power of the immune system to fight cancer. These treatments can either help the immune system attack the cancer directly or stimulate the immune system in a more general way.
Vaccines and checkpoint inhibitors are two types of immunotherapy being tested in prostate cancer. Treatment vaccines are injections that stimulate the immune system to recognize and attack a tumor.
One type of treatment vaccine called sipuleucel-T (Provenge) is approved for men with few or no symptoms from metastatic CRPC.
Immunotherapy: checkpoint inhibitors for prostate cancer
An immune checkpoint inhibitor is a type of drug that blocks proteins on immune cells, making the immune system more effective at killing cancer cells.
Two checkpoint inhibitors, pembrolizumab (Keytruda) and dostarlimab (Jemperli) have been approved for the treatment of tumors, including prostate cancers, that have specific genetic features . Pembrolizumab has also been approved for any tumor that has metastasized and has a high number of genetic mutations .
But relatively few prostate cancers have these features, and prostate cancer in general has largely been resistant to treatment with checkpoint inhibitors and other immunotherapies, such as CAR T-cell therapy .
Research is ongoing to find ways to help the immune system recognize prostate tumors and help immune cells penetrate prostate tumor tissue. Studies are looking at whether combinations of immunotherapy drugs, or immunotherapy drugs given with other types of treatment, may be more effective in treating prostate cancer than single immunotherapies alone.
Targeted radiation therapy and PSMA
Scientists have developed targeted therapies based on PSMA, the same protein that is being tested for imaging prostate cancer. For treatment, the molecule that targets PSMA is chemically linked to a radioactive compound . This new compound can potentially find, bind to, and kill prostate cancer cells throughout the body.
In a recent clinical trial, men with a type of advanced prostate cancer who received a PSMA-targeting drug lived longer than those who received standard therapies . This trial led to FDA approval of the drug, Lu177-PSMA-617 (Pluvicto) , to treat some people with metastatic prostate cancer. Ongoing and planned clinical trials are testing PSMA-targeting drugs in patients with earlier stages of prostate cancer, and in combination with other treatments, including targeted therapies like PARP inhibitors and immunotherapy.
Personalized clinical trials for prostate cancer
Research is uncovering more information about the genetic changes that happen as prostate cancers develop and progress. Although early-stage prostate cancer has relatively few genetic changes compared with other types of cancer, researchers have learned that metastatic prostate cancers usually accumulate more mutations as they spread through the body.
These mutations may make men with metastatic prostate cancers candidates for what are called “basket” clinical trials of new drugs. Such trials enroll participants based on the mutations found in their cancer, not where in the body the cancer arose. In the NCI-MATCH trial , a high percentage of enrolled men with advanced prostate cancer had mutations that could potentially be targeted with investigational drugs.
NCI-Supported Research Programs
Many NCI-funded researchers working at the National Institutes of Health campus, as well as across the United States and world, are seeking ways to address prostate cancer more effectively. Some of this research is basic, exploring questions as diverse as the biological underpinnings of cancer and the social factors that affect cancer risk. And some is more clinical, seeking to translate basic information into improving patient outcomes. The programs listed below are a small sampling of NCI’s research efforts in prostate cancer.
- The Cancer Biomarkers Research Group promotes research on cancer biomarkers and manages the Early Detection Research Network (EDRN) . EDRN is a network of NCI-funded institutions that are collaborating to discover and validate early detection biomarkers.
- Within the Center for Cancer Research , the Prostate Cancer Multidisciplinary Clinic (PCMC) provides comprehensive consultations on diagnosis and treatment options to people with newly-diagnosed prostate cancer.
- The Prostate Specialized Programs of Research Excellence (Prostate SPOREs) are designed to quickly move basic scientific findings into clinical settings. The Prostate SPOREs support the development of new therapies and technologies and studies to better understand how to prevent, monitor, and treat prostate cancer.
- The NCI Cancer Intervention and Surveillance Modeling Network (CISNET) focuses on using modeling to improve our understanding of which men are most likely to benefit from PSA-based screening. CISNET also studies treatment strategies for prostate cancer and approaches for reducing prostate cancer disparities.
- The NCI Genitourinary Malignancies Center of Excellence (GUM-COE) brings together scientists studying genitourinary cancers (GU) from across NCI’s Center for Cancer Research and the Division of Cancer Epidemiology and Genetics, as well as investigators who study GU malignancies in other institutes of NIH. The goal is to provide a centralized resource and infrastructure to accelerate the discovery, development, and delivery of interventions for the prevention, diagnosis, and treatment of these cancers.
- The Research on Prostate Cancer in Men with African Ancestry (RESPOND) study is the largest-ever coordinated research effort to study biological and non-biological factors associated with aggressive prostate cancer in African American men. The study , launched by NCI and the National Institute on Minority Health and Health Disparities in partnership with the Prostate Cancer Foundation, is looking at the environmental and genetic factors related to the aggressiveness of prostate cancer in African American men to better understand why they disproportionally experience aggressive disease.
Clinical Trials
NCI funds and oversees both early- and late-phase clinical trials to develop new treatments and improve patient care. Trials are available for prostate cancer prevention , screening , and treatment .
Prostate Cancer Research Results
The following are some of our latest news articles on prostate cancer research:
- Enzalutamide Gets Added Approval for Prostate Cancer That Hasn’t Spread
- FDA Approves New Initial Treatment Option for Some Metastatic Prostate Cancers
- Is a Genomic Test Better at Finding Aggressive Prostate Cancer?
- Active Surveillance for Low-Risk Prostate Cancer Continues to Rise
- Darolutamide Extends Survival for Some People with Metastatic Prostate Cancer
- Shorter, More Intensive Radiation Safe after Surgery for Prostate Cancer
View the full list of Prostate Cancer Research Results and Study Updates .
- International edition
- Australia edition
- Europe edition
New prostate cancer treatment may be ‘on the horizon’, say scientists
Ability to reverse cancer’s resistance to therapy opens possibility of treatment for late-diagnosed men
Scientists say a new way to treat prostate cancer may be on the horizon after finding it is possible to reverse its resistance to therapy.
More than a million men worldwide are diagnosed with the disease each year. The chances of survival are generally good, particularly if it is diagnosed early. Many can live for decades without symptoms or needing treatment.
But some with advanced forms of prostate cancer find it is able to evade treatment by using their immune system to resist the impact of drugs. Now scientists have discovered a way to stop it being able to do this, opening up the possibility of treatments for men left with little hope.
By blocking the secret messages that cancer cells send to hijack healthy white blood cells, researchers were able to reverse resistance to therapy in a small group of patients. In some, they were able to shrink tumours or halt their growth. The findings were published in Nature.
“This is tremendously exciting and it suggests we have an entirely new way to treat prostate cancer on the horizon,” said Johann de Bono, a professor of experimental cancer medicine at the Institute of Cancer Research and consultant medical oncologist at the Royal Marsden NHS foundation trust.
In a trial led by the ICR, the Royal Marsden and the Institute of Oncology Research in Switzerland, scientists recruited 23 patients with advanced prostate cancer that had stopped responding to hormone therapy.
They were given a combination of AZD5069, an experimental drug that prevents white blood cells from being dragged inside tumours, and enzalutamide, a hormone therapy commonly used to treat prostate cancer.
Of 21 patients who could be evaluated, five (24%) showed evidence of their tumours responding to the combination, the ICR reported.
Their tumours shrunk by more than 30%, they experienced “dramatic decreases” in circulating levels of prostate specific antigen (PSA), a marker often elevated by cancer, or their blood levels of circulating tumour cells dropped, it said.
Patients also showed a drop in the white blood cells targeted by the treatment – myeloid cells – in the blood, and biopsies revealed fewer of them in their tumours.
“It’s hugely rewarding to see our theory proven in a trial of patients with this disease,” said De Bono. “Myeloid cells may be implicated in treatment resistance in a range of cancers, so the impact of this research could be very broad, across multiple cancer types.”
after newsletter promotion
Prof Kristian Helin, chief executive of the ICR, said the results acted as a proof of principle for disrupting cancer and represented a smart new way to attack tumours.
“I look forward to seeing how this work progresses and hope it will pave the way to a new treatment that is beneficial to people with prostate cancer and potentially also many other cancer types.”
The study was funded by Prostate Cancer UK, Cancer Research UK, the Swiss Card Onco grant organisation, the Prostate Cancer Foundation, AstraZeneca, Wellcome and the NIHR Biomedical Research Centre at the Royal Marsden and ICR, with support from the Experimental Cancer Medicine Centres network.
Prostate Cancer UK’s director of research, Dr Matthew Hobbs, said he was extremely excited about the findings.
He said: “Now we want to see pharmaceutical companies working with researchers to develop new drugs based on what we’ve learnt and to test them in larger trials – turning research into reality for men.”
- Prostate cancer
- Cancer research
- Medical research

What is preventive chemotherapy and how effective is it? The Princess of Wales’ treatment explained

Prostate cancer cases worldwide likely to double by 2040, analysis finds
Study offers hope in identifying high-risk prostate cancer patients

Princess of Wales’ diagnosis: cancers in young are rising, but so are survival rates
UK scientists working on breast cancer monitor fitted in bra

Getting fitter can reduce prostate cancer risk by 35%, study finds

UK cancer study shows big fall in death rates since early 1990s

Sussex man doing well a year and a half after new brain cancer treatment

Lock, Stock and Two Smoking Barrels actor Jake Abraham dies aged 56
Drug that could slow womb cancer to be rolled out by NHS in England
Most viewed.
An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
Preview improvements coming to the PMC website in October 2024. Learn More or Try it out now .
- Advanced Search
- Journal List
- Cancers (Basel)

Metastatic Prostate Cancer—A Review of Current Treatment Options and Promising New Approaches
Philip posdzich.
1 Department of Urology, University Hospital Essen, 45147 Essen, Germany
Christopher Darr
Thomas hilser.
2 Department of Internal Medicine (Oncology), University Hospital Essen, 45147 Essen, Germany
Ken Herrmann
3 Department of Nuclear Medicine, University Hospital Essen, 45147 Essen, Germany
Boris Hadaschik
Viktor grünwald, simple summary.
Prostate cancer is the most common tumor in men. Although there have been many new developments in the last few years, metastatic castration resistant prostate cancer remains a deadly disease. This article provides an overview of currently approved treatment options as well as new therapies that are not standard of care yet. All relevant developments from classical androgen deprivation therapy (ADT) to bispecific T-cell engagers (BiTE) are considered.
Androgen deprivation therapy (ADT) alone has been the standard of care for many years in men with metastatic prostate cancer. Due to the limited survival under this monotherapy, many new treatment options have been developed in the last few years. Regarding hormone-sensitive prostate cancer, combination therapies of two or three agents of ADT, androgen receptor signaling inhibitors (ARSI) and chemotherapy have been established and led to a significant benefit in overall survival. Additionally, in patients with metastatic castration-resistant prostate cancer, there are many new therapeutic approaches. Chemotherapy alone has been the standard of care in this situation. In the last years, some new therapeutic options have been developed, which led to an improved survival after progression under chemotherapy. These therapies include ARSI, PARP inhibitors and Lu-PSMA radioligand therapy. The use of a bispecific T-cell engager (BiTE) in this setting is a new promising therapeutic approach, which has not been established as standard of care yet. The role of immunotherapy in prostate cancer is still under investigation. Overall, many new treatment options make prostate cancer therapy a challenging and promising field.
1. Introduction
Prostate cancer is the most common solid cancer in men worldwide. The number of estimated new prostate cancers in the US in 2022 was 268,490. The number of estimated deaths due to prostate cancer is 34,500 [ 1 ]. Surgery and radiotherapy are the mainstay of treatment in localized disease. Androgen-deprivation-therapy (ADT), androgen signaling inhibition (ARSI) and chemotherapy dominate the standard medical treatment in recurrent or metastatic disease. However, the majority of patients acquire castration-resistance, which is associated with a poor prognosis.
The latest advancements in systemic treatments were the addition of Poly ADP-Ribose Polymerase Inhibitor (PARPi) and radio-ligand therapies (RLT) to the armentarium. While check point inhibitors (CPI) have revolutionized anti-cancer treatment in many diseases they were less successful in prostate cancer. A major driver of this immune resistance is derived from the dominance of a non-inflamed “cold” tumor environment in prostate cancer [ 2 ]. However, some activity has been noted for ipilimumab in men with mCRPC who were treated with at least one chemotherapy and had evidence of progression after the discontinuation of ADT [ 3 ], as well as for pembrolizumab in a subgroup of genetically instable prostate cancer patients [ 4 ].
Novel concepts are needed to advance immunotherapies in prostate cancer. Sipuleucel-T indicated that T-cell-based therapies were clinically active in principle and have led to further clinical development. Bispecific T Cell engager is a novel promising anti-cancer treatment modality in non-inflamed cancers, such as prostate cancer. These compounds re-direct T-cells to the tumor environment by targeting a cancer-specific epitope, such as PSMA in prostate cancer, which is linked to a component of the T-cell receptor (TCR). This mechanism recruits T-cells to the tumor milieu by binding to prostate cancer cells, which activates T-cells and enables immunologic anti-tumor response. In this article, we will give an overview of the current therapy standard in mHSPC and mCRPC, and introduce bispecific T-cell-engager as a new mechanism of action to overcome immune resistance.
A review of the Medline database through PubMed was conducted to identify pivotal trials of approved and contemporary medical treatments in prostate cancer. Guideline-recommended regimens were selected and the current EAU Guideline on prostate cancer served as a reference.
3. Current Treatment Options in Metastatic Prostate Cancer
3.1. metastatic hormone-sensitive prostate cancer (mhspc), 3.1.1. prognostic factors.
Several criteria have been established to estimate the prognosis of metastatic hormone-sensitive prostate cancer. Most common criteria are CHAARTED and LATITUDE criteria. These are summarized in Table 1 and Table 2 .
CHAARTED criteria [ 5 ].
LATITUDE Criteria [ 6 ].
The occurrence of “de novo” metastatic hormone-sensitive disease at the time of initial diagnosis was identified as a poor prognostic factor. The median overall survival (OS) was 51.6 months in low volume disease (HR = 1.64; 95% CI 1.16–2.31) and 43.2 months in high volume disease (HR = 2.48, 95% CI 1.83–3.36) compared to the reference group (prior local treatment/low volume disease HR = 1). These results differed substantially from those patients who had local therapy prior to the diagnosis of metastatic disease, i.e., metachronous metastatic prostate cancer. In this population, OS in low volume disease was 92.4 months (HR = 1) and 55.2 months in high volume disease (HR = 1.9, 95% CI 1.31–2.75 [ 7 ].
ADT is the standard treatment approach in mHSPC patients and may consist of Luteinizing hormone-releasing hormone (LHRH) agonists, LHRH antagonists or bilateral orchiectomy. In previous decades, ADT resided as single modality, but this is not the standard of care (SOC) anymore. OS in mHSPC patients treated with ADT alone is about 42 months [ 8 ].
Today, therapies that combine ADT with ARSI, chemotherapy, or both, yielded superior OS results. Single agent ADT has limited value and remains an option in elderly, frail patients, only.
3.1.3. Combination Therapies
The advent of combination therapies has intensified medical treatment and improved OS prognosis in prostate cancer patients with mHSPC. Different components were added to the ADT backbone.
An early set of treatment intensification trials tested the combination of ADT + docetaxel. There were two important trials, which investigated the combination of ADT and Docetaxel.
The STAMPEDE trial included patients with newly diagnosed M1 or N+ disease, locally advanced disease (cT3, cT4, ISUP grade at least 4, PSA at least 40 ng/mL) and patients with relapse after local treatment (PSA at least 4 ng/mL or PSA doubling time under 6 months or PSA level over 20 ng/mL, M or N relapse). In all, 689/724 (95%) of patients in the control arm and 347/362 patients in the Docetaxel arm had no previous treatment. A total of 1184 patients received the standard of care (ADT) and 592 patients received ADT + docetaxel. The median OS in the ADT group was 43.1 months, the estimated 5-year OS was 37% (CI 34–41%). In contrast, the median OS in patients who received ADT + Docetaxel was 59.1 months, the estimated 5-year OS was 49% ( p = 0.003, HR = 0.81, 95% CI 0.69–0.95) [ 9 ].
The second Important trial was called CHAARTED, which also investigated the impact of ADT + docetaxel in patients with mHSPC and ECOG performance score 0–2. A distinction was made between high volume and low volume disease. In the overall patient population, OS was 57.6 months in the ADT + docetaxel group in contrast to 47.2 months in the ADT alone group (HR = 0.72, 95% CI 0.59 to 0.89; p = 0.0018). Subgroups of patients with high and low volume disease achieved different treatment effects. While ADT + docetaxel vs. ADT achieved a superior OS of 51.2 vs. 34.4 months (HR = 0.63, 95% CI 0.50 to 0.79; p < 0.001) in high volume disease, there was no benefit in patients with low volume disease in longterm follow-up (HR = 1.04, 95% CI 0.70 to 1.55; p = 0.86) [ 10 ].
As a result, OS in newly diagnosed metastatic hormone-sensitive prostate cancer is superior for ADT + docetaxel than ADT alone, but that effect of chemotherapy is restricted to high volume disease only.
3.1.4. ADT + Androgen Receptor Signaling Inhibitors (ARSI)
Abiraterone acetate: selective inhibitor of steroid 17α-hydroxylase (cyp17a1).
In this section, we will discuss the impact of three different androgen receptor signaling inhibitors in addition to ADT.
The STAMPEDE trial also investigated the impact of Abiraterone Acetate (AA), a selective inhibitor of the enzyme steroid 17α-hydroxylase (CYP17A1) and prednisone in addition to ADT. Inclusion criteria were already mentioned above. In all, 957 received ADT alone, 960 patients were treated with ADT and Abiratrone Acetate + prednisone (AAP). There was a significant benefit in OS (HR = 0.63, 95% CI 0.52 to 0.76; p < 0.001) in patients who received the combination therapy which corresponded to 3-year OS rates 83% vs. 76%. Metastatic status at time of randomization had no significant impact on treatment effect ( p = 0.37) [ 11 ].
In the LATITUDE trial, ADT + Placebo ( n = 597 pts.) was compared to ADT + AAP ( n = 602 pts.). Only patients with high-risk newly diagnosed metastatic disease, ISUP grade > 4, at least three bone lesions or measurable visceral metastases were included. The combination of ADT + AAP reported significant OS improvement (HR = 0.62, 95% CI 0.51–0.76; p < 0.001) when compared to ADT alone. Three year OS rates were: 66% in ADT + AAP vs. 49% in ADT + Placebo group [ 6 ].
In summary, the combination therapy of ADT + AAP provided a significant survival benefit over ADT monotherapy.
Enzalutamide: A Competitive Androgen Receptor Blocker
The ENZAMET trial included mHSPC patients (with distant metastases (M1) and ECOG score 0–2). 1125 men (588 with high volume disease, 537 with low volume disease) were 1:1 randomized to receive enzalutamide + ADT or ADT + standard nonsteroidal antiandrogen drug (bicalutamide, nilutamide, flutamide). Overall survival favored ADT + enzalutamide (HR = 0.67, 95% CI 0.52–0.86; p = 0.002) and 3 year OS rate were 80% in the Enzalutamide group and 72% for ADT alone. Regarding the volume of disease, the proportion alive after 36 months was 0.82 (0.75 to 0.87) in the control group vs. 0.90 (0.84 to 0.93) in the Enzalutamide group and low volume disease. In high volume disease, proportion alive after 36 months was 0.64 (0.58 to 0.70) in the control group and 0.71 (0.64 to 0.76) in the Enzalutamide group. There was no statistically significant impact of volume of disease ( p = 0.14) [ 12 ].
Apalutamide: Inhibitor of the Ligand-Binding Domain of the Androgen Receptor
In the TITAN study, apalutamide + ADT ( n = 525) was compared to ADT + Placebo ( n = 527) in patients with mHSPC. A total of 94 of 525 patients in the Apalutamide group and 79 of 527 in the placebo group had previous treatment for localized disease. The other patients were de novo metastasized. The study reported superior OS in favor for the combination arm (HR = 0.67, 95% CI 0.51–0.89; p = 0.005). After 24 months, there was an overall survival of 82.4% in the Apalutamide group, but only 73.5% OS in the Placebo group [ 13 ] In the final analysis after 405 deaths, it was shown that apalutamide decreased the risk of death by 35% (HR = 0.65; 95% CI, 0.53 to 0.79; p < 0.0001) [ 14 ].
The results of these trials indicated that the combination therapy of ADT and ARSI improved OS in mHSPC patients than ADT alone. Both patient groups, high volume disease and low volume disease had a benefit from combination therapy.
3.1.5. Triple Combinations (ADT + ARSI + Docetaxel)
In this section, triple combinations in the treatment of mHSPC will be discussed.
Darolutamide is a competitive androgen receptor inhibitor and reported improved metastasis free survival and overall survival in non-metastatic CRPC patients. The principal activity paved the way for further testing in an earlier setting. The ARASENS trial investigated the combination of darolutamide + ADT + docetaxel ( n = 651) compared to placebo + ADT + docetaxel ( n = 655). The primary analysis at first data cut-off showed that the risk of death was reduced by 32.5% in the triple-therapy arm when compared to the ADT + docetaxel doublet [ 15 ].
PEACE-1 is a complex study and consisted of four arms. At this point, only the combination therapy of ADT + docetaxel with or without abiraterone acetate and prednisone (AAP) were considered for analyses. The median rPFS was prolonged by 2.5 years in patients who received the AAP containing triplet (HR = 0.5, 95% CI 0.40–0.62; p < 0.0001). Furthermore, there was a 25% reduction in the risk of death in patients who received the triplet (HR = 0.75, 95% CI 0.59–0.95); p = 0.017). The addition of Abiraterone improved the median OS from 4.72 years (SOC) to 5.72 years (SOC+abiraterone) (HR = 0.82, 95% CI 0.69–0.98; p = 0.030). Regarding patients with high tumor volume, there was a median survival benefit of 1.5 years in patients who were treated with the triplet (HR = 0.72, 95% CI 0.55–0.95; p = 0.019) [ 16 ].
As a result, triple therapies show a promising survival benefit in mHSPC patients, especially in those with high tumor burden. There are no comparisons between the triple and ADT + ARSI doublets available.
Table 3 provides a summary of the mentioned treatment options in mHSPC:
List of treatment options and survival rates in mHSPC.
3.2. Metastatic Castration Resistant Prostate Cancer (mCRPC)
3.2.1. firstline treatment in mcrpc.
First-line treatment in men with mCRPC differ. Most trials explored the role of treatments after the failure of ADT alone.
Docetaxel was reported to improve OS compared to Mitoxantrone in the SWOG 99–16 trial. Docetaxel/Estramustine (every 3 weeks 60 mg/m 2 ) was compared to Mitoxantrone and prednisone (every 3 weeks 12 mg/m 2 ). Patients who received docetaxel had an overall survival of 17.5 months whereas patients who were treated with Mitoxantrone had an OS of only 15.6 months ( p = 0.02; HR = 0.80; 95% CI 0.67–0.97). There was a significant difference in rPFS:6.3 months (Docetaxel) vs. 3.2 months (Mitoxantrone) ( p < 0.001) [ 17 ].
The efficacy of abiraterone acetate before chemotherapy was shown in the placebo controlled COU-AA-302 trial. Patients who received abiraterone acetate + prednisone had superior OS than those in placebo + prednisone group (34.7 vs. 30.3 months HR 0.81, p = 0.0033). There was also a significant improvement in rPFS favoring AAP (16.5 months vs. 8.3 months; p < 0.0001) [ 18 ].
PREVAIL investigated the therapeutic benefit of enzalutamide in comparison to placebo in mCRPC in chemotherapy-naïve patients. Patients treated with enzalutamide had an OS of 32.4 months compared to 30.2 months in placebo group (HR = 0.71, 95% CI 0.60–0.84, p < 0.001). rPFS was significantly longer in patients who received enzalutamide (20.0 months vs. 5.4 months (HR = 0.186, 95% CI 0.15–0.23; p < 0.0001) [ 19 ].
The PROPEL trial investigated the combination of Olaparib + Abiraterone vs. Placebo + Abiraterone. This trial will be mentioned in detail in section “Molecular Therapies”.
Firstline options in CRPC are summarized in Table 4 .
Firstline options in CRPC.
3.2.2. Options after Pretreatment in mCRPC
For mCRPC patients who have already been treated with docetaxel, there are some therapeutic options, which will be listed below.
Cabazitaxel is a novel taxane with activity in docetaxel resistant CRPC. In the TROPIC 2013 trial, Cabazitaxel + prednisone showed a benefit in 2 year OS in comparison to mitoxantrone + prednisone (OS > 2 years in 15.9% (60/378) vs. 31/377 (8.2%). (odds ratio 2.11; 95% CI 1.33–3.33) [ 20 ].
The value of AAP in treatment of mHSPC has already been mentioned above. Additionally, in mCRPC it is a possible therapy option after docetaxel. In COU AA 301 study, AAP was compared to placebo/prednisone and led to an OS of 15.8 months in AAP group vs. 11.2 months in the placebo arm. ( p < 0.0001, HR = 0.74, 95% CI: 0.64–0.86). Median rPFS was 5.6 months (5.6–6.5) in AAP group and 3.6 months (2.9–5.5) in control group (HR = 0.66, 0.58–0.76; p < 0·0001) [ 21 ].
Enzalutamide has been mentioned above. In the AFFIRM trial, patients who received enzalutamide after docetaxel had a significantly longer OS than those who received placebo (OS 18.4 vs. 13.6 months; ( p < 0.001, HR = 0.63; 95% CI: 0.53–0.75)). There was also a statistically significant difference in rPFS: 8.3 months vs. 2.9 months ( p < 0.001, HR = 0.63; 95% CI: 0.53–0.75) that favored enzalutamide [ 22 ].
In patients with two or more symptomatic bone metastases and no visceral metastases, alpharadin (Radium 223) is also a possible treatment option. Besides symptom relief in bone metastases, it also led to a longer OS in ALSYMPCA (Radium 223 vs. Placebo in previous or no previous docetaxel). OS in patients who received Radium 223 was 14.9 months vs. 11.3 months in placebo group ( p = 0.002, HR = 0.61; 95% CI: 0.46–0.81) The most common hematologic AE in the Radium-223 group was anemia (187/600 pts.; 31%) whereas bone pain (300/600 pts.; 50%) and nausea (213/600 pts.; 36%) where most common nonhematologic AEs. [ 23 ].
In the CARD study, Cabazitaxel was compared to Abiraterone or Enzalutamide in patients with mCRPC who were treated with Doxetaxel and Abiraterone or Ezalutamide. In all, 255 patients were randomized in two groups: one group received Cabazitaxel (129 pts.), the other group received Abiraterone or Enzalutamide depending on which they had not received yet (126 pts.). The median overall survival was 13.6 months in the Cabazitaxel group and 11.0 months in the Abiraterone/Enzalutamide group (HR for death = 0.64; 95% CI, 0.46 to 0.89; p = 0.008). The median progression-free survival was 4.4 months in Cabazitaxel group vs. 2.7 months in the other group. (HR for progression or death =0.52; 95% CI, 0.40 to 0.68; p < 0.001). As a result, Cabazitaxel resulted in longer overall survival and progression-free survival [ 24 ].
Options after pretreatment are summarized in Table 5 .
Options after pretreatment in mCRPC.
4. Molecular Therapy
The incorporation of molecular diagnostics deepened our understanding of putative therapeutic avenues in mCRPC. BRCA 1 or 2 regulate homologous recombination (HR) and loss in BRCA function may occur as germline or sporadic alteration. BRCA deficient cancers are explicitly susceptible to PARP inhibitors. The clinically most advanced PARP inhibitor in mCRPC is olaparib. The PROfound study compared olaparib with AAP or enzalutamide in patients with alterations in HR repair (HRR) deficient CRPC after treatment with a new hormonal agent. BRCA 1 alteration was found in 8/256 pts in Olaparib group (3%) and 5/131 pts. in control group (4%). BRCA 2 alteration was found in 81/256 pts (32%) in the Olaparib group and 47/131 (36%) in the control group. In patients with at least one alteration in BRCA1, BRCA 2 or ATM, Olaparib achieved a superior rPFS (7.39 vs. 3.55 mo. (HR = 0.34; 95% CI: 0.25–0.47; p < 0.0001)) and OS (18.5 mo vs. 15.1 mo. hazard ratio for death = 0.64; 95% CI, 0.43 to 0.97; p = 0.02). In the overall population, the median OS at interim analysis was 17.5 months (Olaparib group) and 14.3 months (control group) (hazard ratio for death = 0.67; 95% CI, 0.49 to 0.93). Patients with BRCA alterations derived the largest benefit, which led to a restricted label in Europe. However, other HRR alterations seem to select for PARPi-sensitivity, but subgroups remain small and their predictive strength remains vague [ 25 ].
In the PROpel study, 796 patients with mCRPC were randomized to receive Olaparib + Abiraterone ( n = 399) vs. Placebo and Abiraterone ( n = 397). In an interim analysis, rPFS was longer in Olaparib + Abiraterone group irrespective of HRR status (24.8 vs. 16.6 months; HR = 0.66, 95% confidence interval [CI] 0.54–0.81; p < 0.0001). Data on OS were immature at interim analysis [ 26 ].
Based on these data, BRCA testing has entered the clinical routine in mCRPC.
Alterations of the PI3 kinase/AKT signaling pathway are common in CRPC. PTEN is a suppressor of this signaling pathway and its loss is frequently found in CRPC. PTEN loss is associated with an aggressive clinical course and subject for pharmacological intervention in late stage mCRPC. Ipatasertib is an AKT inhibitor and its clinical efficacy was tested in a pivotal trial. Patients with previously untreated asymptomatic or mildly symptomatic mCRPC were eligible. The combination of Ipatasertib and AAP showed a benefit in rPFS in patients with PTEN loss in comparison to AAP + placebo (18.5 months vs. 16.5 months p = 0.0335, HR = 0.77, 95% CI: 0.61–0.98). There was no significant benefit on PFS in the intention to treat population (16.6 months (Placebo) vs. 19.2 months (HR = 0.84 [95% CI 0·71–0.99]; p = 0.043) [ 27 ].
A new therapeutic modality was introduced with the development of systemic PSMA-targeted radio ligand therapy (RLT) in mCRPC. Lutetium-177-PSMA-617 (LU-PSMA) is a small molecule that binds specifically to PSMA, which enables ß particle therapy to adjacent tumor cells in CRPC. A positive diagnostic 68-Gallium PSMA PET scan is a prerequisite to select suitable patients for this molecular therapy.
The VISION trial tested LU-PSMA in previously treated mCRPC patients who were not candidates for chemotherapy. LU-PSMA showed a significant benefit in rPFS (8.7 vs. 3.4 mo. ( p < 0.001; HR = 0.40; 99.2% CI: 0.29–0.57)) and OS (15.3 vs. 11.3 mo. ( p < 0.001; HR = 0.62; 95% CI: 0.5–0.74)) in comparison to standard of care (best supportive care, which consisted of hormonal therapy, denosumab, bisphosphonates, radiation therapy or glucocorticoids) [ 28 ].
Another important trial which proved the effectiveness of PSMA radioligand therapy was TheraP. It included previously treated mCRPC patients who were deemed fit for chemotherapy and patients were randomized to receive LU-PSMA or Cabazitaxel. Patient selection was more stringent and permitted only patients with a match of PSMA- and FDG-PET activity. LU-PSMA RLT (up to six cycles; n = 98 pts.) was compared to Cabazitaxel (up to 10 cycles; n = 85 pts.). PSA response (defined as reduction of at least 50% from baseline), the primary endpoint, was shown in 66/98 (67%) pts who received PSMA RLT, but only 37/85 pts. (43%) who received Cabazitaxel ( p < 0.001). Grade 3–4 adverse events occurred in 32 (33%) patients in the PSMA RLT group vs. 45 (53%) in the Cabazitaxel group.
Overall, PSMA RLT showed improved efficacy and lower risk of grade 3–4 adverse events when compared to Cabazitaxel. However, the trial did not indicate major differences in OS in this trial and underlines the value of LU-PSMA in direct comparison to Cabazitaxel in mCRPC patients, which is considered a life-prolonging therapy [ 29 ].
Both BRCA 1/2 mutations and PSMA-positivity were predictive markers for treatment benefit. The molecular therapies are summarized in Table 6 .
Molecular therapies.
The most common adverse events related to molecular therapies are shown in Table 7 .
Most common adverse events related to molecular therapy.
4.1. Immunotherapy
Until now, immunotherapy has not been of great importance in the treatment of prostate cancer and did not improve clinical outcomes significantly. Current trials test the role of ICI in specific subgroups: KEYNOTE- 641 is a Phase 3, randomized, double-blind, placebo-controlled trial, which investigates Pembrolizumab/Placebo in combination with Enzalutamide in mCRPC patients who were not treated with abiraterone or progressed on Abiraterone and did not receive chemotherapy [ 30 ]. Keynote-921 is a Phase 3, randomized, double-blind, placebo-controlled trial which investigates pembrolizumab/placebo in combination with docetaxel and prednisone in mCRPC patients who have received novel hormonal agents but no chemotherapy before [ 31 ]. On 3 August 2022, the sponsor announced that the study did not meet its primary endpoints (improvement in overall survival and rPFS). KEYNOTE-991 is a Phase 3 trial to investigate the role of Pembrolizumab in combination with Enzalutamide and ADT versus Placebo in combination with Enzalutamide and ADT in mCRPC patients who did not receive novel hormonal agents before. The results of these trials are not available until 2025/2026 and could reform the rule of immunotherapy in prostate cancer [ 32 ]. Keynote-365 is a multicohort phase Ib/II study which tests pembrolizumab in combination with Olaparib (Cohort A), Docetaxel and Prednisone (Cohort B), Enzalutamide (Cohort C). In the results published so far, PSA response was reported in 9% of patients in cohort A, 28% in cohort B and 22% in cohort C. The final results of this study are not yet available [ 33 ].
4.2. Cell Based Immunotherapy
So far, cell based immunotherapy has not found its place in the treatment landscape of mCRPC. This could change in the future due to ongoing developments.
A therapy approach which was developed several years ago is supileucel-T. This is an active cell based autologous immunotherapy. Peripheral blood mononuclear cells are activated ex vivo with PA2024, a recombinant fusion protein of prostate antigen, which causes immune cell activation. Ultimately, mCRPC patients with an expected survival of at least 6 months received supileucel-T in a phase III trial ( n = 341 pts.). In all, 171 patients received placebo as a control group. The OS in the supileucel-T group was 25.8 months and 21.7 months in the placebo group. (HR for death in the sipuleucel-T group = 0.78; 95% confidence interval [CI], 0.61 to 0.98; p = 0.03) [ 34 ]. Although showing principal activity, supileucel-T is not available in routine practice anymore.
5. Vaccination
Vaccination against prostate cancer is not established in clinical practice. Nevertheless, there are studies that provided promising data. As an example, PROSTVAC, a PSA recombinant vaccinia vector, showed promising activity in mCRPC patients. Median OS was 8.5 months longer in pts who received PROSTVAC than in the control group ( p = 0.0061) [ 35 ]. However, confirmatory trials were negative and did not show a benefit in OS [ 36 ].
6. Bispecific T-Cell Engager
The most advanced bispecific T-cell engager in the therapy of prostate cancer is PSMA Bite. PSMA is expressed in prostate cancer cells and metastases and can be used to specifically target therapies to prostate cancer cells, such as for LU-PSMA radioligand therapy [ 28 ].
PSMA Bite is a new experimental treatment for mCRPC patients. PSMA Bite is a bispecific CD3 and PSMA antibody construct, which re-directs and activates T-cells to PSMA expressing cells.
Bispecific T-cell engager (BiTE) is already established in the treatment of other malignancies. Blinatumumab was the first approved BITE therapy. It is a bispecific monoclonal antibody construct that causes CD3 positive T-cells to recognize and target CD 19 positive B-cells. Blinatumumab is approved for patients with refractory or relapsed precursor B-ALL. Compared to chemotherapy, blinatumumab showed a survival benefit in patients with pretreated B-ALL: Median OS in the blinatumomab group was 7.7 months compared to 4.0 months in the chemotherapy group (HR = 0.71; 95% CI 0.55 to 0.93; p = 0.01) [ 37 ].
Based on promising data from the treatment of hematological diseases, the suitability of bite molecules for the treatment of solid tumors is currently being investigated.
A Phase I study of pasotuximab (PSMA Bite) tested its tolerability and activity in mCRPC. It included 68 patients in two cohorts of subcutaneous (s.c.) or intravenous (i.v.) application. These patients were pretreated with at least one taxane regimen and refractory to AAP or enzalutamide. Next, to evaluate the maximum tolerated dose in both cohorts, PSA response was investigated. In the subcutaneous cohort, every patient developed anti-drug antibodies, which led to the premature discontinuation of the s.c. application cohort.
PSA decline was −24.7% in pts. with s.c. treatment. In the i.v. group, median best PSA change was −22.0, −37.7 and −54.9% in 20, 40 and 80 µg/d dose cohorts. One patient in i.v. cohort had <50% PSA reduction for 50 weeks and stable disease for 337 days. Another patient in the i.v. cohort had nearly complete regression of lymphnode and bone metastases in PSMA-PET CT. PSA progression in long-term responders in i.v. cohort occurred after 11–17 months and indicates the principle and dose-dependent activity of this new modality. Most common adverse events in both cohorts were fever (81% in s.c. and 94% in i.v. cohort), injection site reaction in the s.c. cohort (24/31; 77%), chills (23% in s.c. and 69% in the i.v. cohort) and fatigue (36% in the s.c. and 31% in the i.v. cohort). Treatment-emergend AEs occurred in both cohorts. Most common were anemia (39%) in the s.c. cohort and decreased lymphocyte count (44%) and infections (31%) in the i.v. cohort [ 38 ]. Further trials to investigate the value and safety of PSMA Bite are ongoing.
Other targets than PSMA may be used for bispecific T-cell engager, such as Glypican-1 [ 39 ] and ADAM 17 (disintegrin and metalloproteinase 17) [ 40 ] or STEAP-1 [ 41 ], which are under investigation.
Overall, bispecific T-cell engager ( Figure 1 ) is a promising new therapy option with early signs of clinical activity. Of course, further clinical studies are necessary before they are ready for prime time, but early clinical trials are promising.

Bispecific T-cell engager. Modified according to Strohl et al. [ 42 ].
7. Conclusions
Prostate cancer is the most common solid cancer in men. In recent years, there have been major advances in the treatment of metastatic prostate cancer, which have pushed frontiers of survival expectations to new levels. ADT alone is not enough anymore for men with metastatic HSPC. Men with BRCA 1/2 alterations and with PSMA-positive cancers benefit from targeted treatment with PARPi or Lu-PSMA, respectively. However, castration resistant prostate cancer remains a deadly disease and new therapies are needed. The advent of molecular therapies, such as RLT or PARPi, advanced the field more recently and early clinical trials indicate promising new therapeutic approaches, which includes immunotherapies.
Funding Statement
This research received no external funding.
Author Contributions
Writing, original manuscript preparation: P.P.; editing and review: C.D., T.H., K.H. and M.W.; supervision: B.H. and V.G. All authors have read and agreed to the published version of the manuscript.
Conflicts of Interest
P.P. has no conflicts of interest; C.D. Travels Fees and Advisory Board from Janssen-Cilag; T.H. has no conflicts of interest; M.W. has no conflicts of interest; B.H. has had advisory roles for ABX, AAA/Novartis, Astellas, AstraZeneca, Bayer, Bristol Myers Squibb, Janssen R&D, Lightpoint Medical, Inc., and Pfizer; has received research funding from Astellas, Bristol Myers Squibb, AAA/Novartis, German Research Foundation, Janssen R&D, and Pfizer; and has received compensation for travel from Astellas, AstraZeneca, Bayer and Janssen R&D. KH reports personal fees from Bayer, personal fees and other from Sofie Biosciences, personal fees from SIRTEX, non-financial support from ABX, personal fees from Adacap, personal fees from Curium, personal fees from Endocyte, grants and personal fees from BTG, personal fees from IPSEN, personal fees from Siemens Healthineers, personal fees from GE Healthcare, personal fees from Amgen, personal fees from Novartis, personal fees from ymabs, all outside the submitted work. VG: Receipt of grants/research support: AstraZeneca, Novartis, BMS, MSD, Ipsen, Pfizer; receipt of honoria or consultation fees: AstraZeneca, BMS, Novartis, Apogepha, Ipsen, EISAI, MSD, MerckSerono, Roche, EUSAPharm, Janssen, Nanobiotix, ONO Pharmaceutical, Debiopharm; stock shareholder: AstraZeneca, BMS, SeaGen, MSD, GenMab; travel support: AstraZeneca, BMS, MerckSerono.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
- Alzheimer's disease & dementia
- Arthritis & Rheumatism
- Attention deficit disorders
- Autism spectrum disorders
- Biomedical technology
- Diseases, Conditions, Syndromes
- Endocrinology & Metabolism
- Gastroenterology
- Gerontology & Geriatrics
- Health informatics
- Inflammatory disorders
- Medical economics
- Medical research
- Medications
- Neuroscience
- Obstetrics & gynaecology
- Oncology & Cancer
- Ophthalmology
- Overweight & Obesity
- Parkinson's & Movement disorders
- Psychology & Psychiatry
- Radiology & Imaging
- Sleep disorders
- Sports medicine & Kinesiology
- Vaccination
- Breast cancer
- Cardiovascular disease
- Chronic obstructive pulmonary disease
- Colon cancer
- Coronary artery disease
- Heart attack
- Heart disease
- High blood pressure
- Kidney disease
- Lung cancer
- Multiple sclerosis
- Myocardial infarction
- Ovarian cancer
- Post traumatic stress disorder
- Rheumatoid arthritis
- Schizophrenia
- Skin cancer
- Type 2 diabetes
- Full List »
share this!
April 10, 2024
This article has been reviewed according to Science X's editorial process and policies . Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
New insight into combating drug-resistant prostate cancer
by University of Eastern Finland
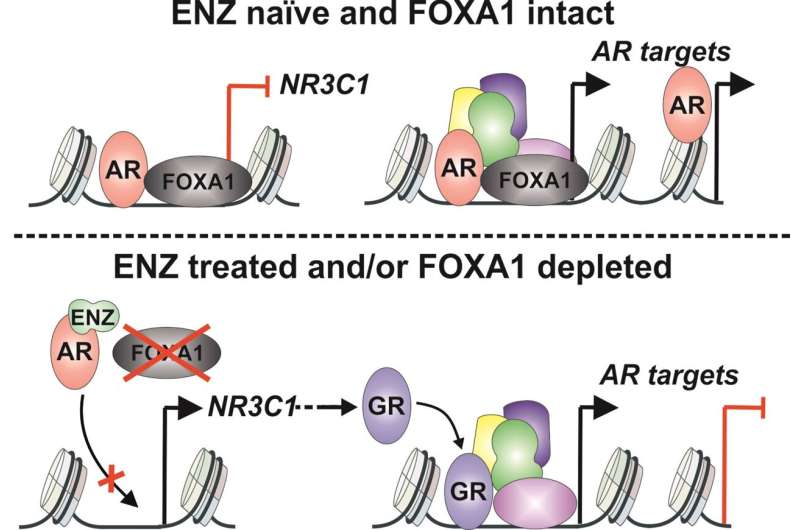
Recent research from the University of Eastern Finland sheds light on the significance of the glucocorticoid receptor in drug-resistant prostate cancer, showing that the development of drug resistance could be prevented by limiting the activity of coregulator proteins.
Glucocorticoids regulate vital biological processes by affecting gene encoding through a DNA-binding transcription factor, namely the glucocorticoid receptor. The activity of the glucocorticoid receptor is made extensive use of in medicine because glucocorticoids have a strong anti-inflammatory effect.
For this reason, synthetic glucocorticoids are one of the most prescribed drugs in the world. They are used to treat inflammatory diseases , such as rheumatoid arthritis , and as adjuvant therapy for cancer patients to alleviate the side effects of cancer therapy. In blood cancer , glucocorticoids are important drugs that limit the growth of cancer cells.
However, recent studies have shown that the glucocorticoid receptor also has an oncogenic, or cancer-promoting, effect in cancers like breast and prostate cancer . In prostate cancer, the glucocorticoid receptor can replace the activity of the androgen receptor, which is the main oncogenic factor in this cancer, when its activity is inhibited by drug therapy. Thus, glucocorticoids help prostate cancer develop resistance to drug therapy.
"Due to these drug resistance and cancer-promoting effects, it is important to study how the glucocorticoid receptor functions on the cellular and molecular level in cancer," Academy Research Fellow, Docent Ville Paakinaho of the University of Eastern Finland notes.
The Paakinaho Lab has published two recent genome-wide deep sequencing studies on the subject. The first, published in Nucleic Acids Research , explored how the glucocorticoid receptor replaces the androgen receptor on the molecular level.
"This study showed that the glucocorticoid receptor can only use regulatory regions that are already active in prostate cancer cells," says Doctoral Researcher Laura Helminen of the University of Eastern Finland.
In other words, glucocorticoid receptor-mediated drug resistance emerges through these regulatory regions, and by affecting the activity of these areas, the harmful effects of glucocorticoids in prostate cancer could be prevented.
Bioinformatics analyses indicated the pioneer transcription factor FOXA1 as one possible target. FOXA1 is known to have cancer-promoting properties, which is why the researchers assumed that inhibiting its activity would limit the development of glucocorticoid receptor-mediated drug-resistant prostate cancer. Surprisingly however, the effect was exactly the opposite: inhibiting the activity of FOXA1 further increased the activity of the glucocorticoid receptor—and the development of drug resistance.
This is because FOXA1 was found to be involved in the silencing of the glucocorticoid receptor gene, and this is what increased its activity when FOXA1 was inhibited.
"Research often reveals the unexpected, and that's part of its charm," Paakinaho says.
The activity of the glucocorticoid receptor in regulatory regions can, however, be influenced in drug-resistant prostate cancer through an alternative pathway. Coregulator proteins were identified as an alternative target through which the glucocorticoid receptor affects the regulation of gene expression. These proteins include EP300 and CREBBP. Several pharmaceutical companies are developing small-molecule inhibitors targeting these proteins, and some are already being studied in patients.
In another study by the Paakinaho Lab, the researchers explored ways to inhibit glucocorticoid receptor-mediated effects by inhibiting coregulator proteins. These findings were reported in Cellular and Molecular Life Sciences .
"Silencing the EP300 and CREBBP proteins with a small-molecule inhibitor clearly prevented the activity of the glucocorticoid receptor in prostate cancer cells," Project Researcher Jasmin Huttunen of the University of Eastern Finland says.
This allowed the growth of drug-resistant prostate cancer cells to be inhibited. Furthermore, the researchers found that silencing EP300 and CREBBP also effectively inhibited the activity of the androgen receptor especially in prostate cancer cells that have an amplification of the androgen receptor gene. This amplification is found in up to half of patients with advanced prostate cancer.
Surprisingly, the EP300 and CREBBP inhibitor also inhibited the activity of FOXA1, while still preserving its ability to silence the expression of the glucocorticoid receptor gene. By using the EP300 and CREBBP inhibitor, it was possible to block the activity of FOXA1 without the development of glucocorticoid receptor-mediated drug resistance .
Ultimately, inhibiting the activity of both the androgen and the glucocorticoid receptor was found to be primarily due to the limitation of FOXA1 activity. The study suggests that treatment targeting coregulator proteins could also be effective in untreated prostate cancer.
Jasmin Huttunen et al, EP300/CREBBP acetyltransferase inhibition limits steroid receptor and FOXA1 signaling in prostate cancer cells, Cellular and Molecular Life Sciences (2024). DOI: 10.1007/s00018-024-05209-z
Explore further
Feedback to editors

Case study of 4-year-old with Down syndrome and sleep apnea suggests procedure can be effective at young ages
2 hours ago

Study finds esketamine injection just after childbirth reduces depression in new mothers
7 hours ago

A new screening protocol can detect aggressive prostate cancers more selectively
9 hours ago
How a new drug prototype regenerates lung tissue

Why some people with rheumatoid arthritis have pain without inflammation
10 hours ago


Researchers show chemical found naturally in cannabis may reduce anxiety-inducing effects of THC
11 hours ago
'Virtual biopsy' lets clinicians analyze skin noninvasively
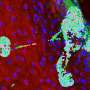
Research team discovers new way to generate human cartilage

Filling in genomic blanks for disease studies works better for some groups than others

Researchers find new origin of deep brain waves
12 hours ago
Related Stories
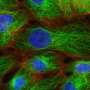
Study shows clinical benefit of a new way of treating advanced ER+ breast cancer
Feb 8, 2024

Neuroendocrine prostate cancer could be restored to a state that responds to treatment
Feb 2, 2024
Acetylation: A timekeeper of glucocorticoid sensitivity
Mar 26, 2024

Cleveland clinic researchers identify new drug target for treating aggressive prostate cancer
May 26, 2021

Potential new drug targets may enhance the treatment of prostate cancer
Aug 18, 2021
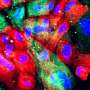
Research team develops new screening assay for drugs targeting prostate cancer
Apr 6, 2022
Recommended for you
Study suggests liquid biopsy could detect and monitor aggressive small-cell lung cancer

Newly found genetic variant defends against Alzheimer's disease
15 hours ago
Researchers identify protein that controls CAR T cell longevity

Researchers explore role of androgens in shaping sex differences
Let us know if there is a problem with our content.
Use this form if you have come across a typo, inaccuracy or would like to send an edit request for the content on this page. For general inquiries, please use our contact form . For general feedback, use the public comments section below (please adhere to guidelines ).
Please select the most appropriate category to facilitate processing of your request
Thank you for taking time to provide your feedback to the editors.
Your feedback is important to us. However, we do not guarantee individual replies due to the high volume of messages.
E-mail the story
Your email address is used only to let the recipient know who sent the email. Neither your address nor the recipient's address will be used for any other purpose. The information you enter will appear in your e-mail message and is not retained by Medical Xpress in any form.
Newsletter sign up
Get weekly and/or daily updates delivered to your inbox. You can unsubscribe at any time and we'll never share your details to third parties.
More information Privacy policy
Donate and enjoy an ad-free experience
We keep our content available to everyone. Consider supporting Science X's mission by getting a premium account.
E-mail newsletter
Skip to Content
- Conquer Cancer
- ASCO Journals
- f Cancer.net on Facebook
- t Cancer.net on Twitter
- q Cancer.net on YouTube
- g Cancer.net on Google
Types of Cancer
- Navigating Cancer Care
- Coping With Cancer
- Research and Advocacy
- Survivorship
Prostate Cancer: Latest Research
ON THIS PAGE: You will read about the scientific research being done to learn more about prostate cancer and how to treat it. Use the menu to see other pages.
Doctors are working to learn more about prostate cancer, ways to prevent it, how to best treat it, and how to provide the best care to people diagnosed with this disease. The following areas of research may include new options for patients through clinical trials. Always talk with your doctor about the best diagnostic and treatment options for you.
Finding causes of prostate cancer. Researchers continue to explore the link between nutrition and other lifestyle factors and the development of prostate cancer.
Early detection. Researchers are trying to develop a better prostate-specific antigen (PSA) test, either a more specific and precise test or a different test. With improved testing, more healthy people could be screened for prostate cancer, so more prostate cancers could be found and treated early.
Genomic tests. Genomics is the study of how genes behave. Genomic tests look at the genes in prostate cancer to help predict how quickly the cancer may grow and spread. The information from these tests can help the cancer care team make decisions about the treatment plan, such as whether active surveillance is an option for those with low-risk prostate cancer or by helping the health care team make a prognosis after surgery and choose the best adjuvant treatments. Some of the genomic tests available now include Decipher, Oncotype DX, ProstaVysion, and the Prolaris Test.
The National Comprehensive Cancer Network (NCCN) recently updated their guidelines to include details about genomic testing in prostate cancer. They recommend that people with metastatic castration-resistant prostate cancer receive testing for inherited and tumor mutations, which could help direct treatment.
Learn more about ASCO recommendations for genomic tests for prostate cancer in the Diagnosis section.
Advanced imaging scans. Research is ongoing to use different molecules in PET-CT scans (positron-emission tomography, computed tomography; see Diagnosis ) to gather important information about a prostate cancer diagnosis, such whether there is distant spread (metastasis).
Improved surgical techniques. Better techniques for nerve-sparing surgery can decrease the risk of urinary and sexual side effects for people who need a radical prostatectomy.
Shorter radiation therapy schedules. With better, more precise external-beam radiation therapy, researchers are exploring much shorter and more convenient treatment schedules. Instead of 40 treatments, researchers are evaluating using 28, 12, or 5 treatments instead.
Tests to evaluate the success of treatment. Research continues to evaluate biomarkers that are found in the blood. These biomarkers can help determine the effectiveness of a treatment and be used to better assess the cancer’s response to treatment. Blood tests measuring circulating tumor cells (CTCs) are 1 such test. CTCs are cells that have broken free from the tumor.
Improved therapy for advanced prostate cancer. Researchers are exploring different treatment options for advanced prostate cancer in clinical trials, including special targeted drugs, chemotherapy, hormonal therapy, immunotherapy, and combinations of different types of therapies.
Palliative care/supportive care. Clinical trials are underway to find better ways of reducing symptoms and side effects of current prostate cancer treatments to improve comfort and quality of life for patients.
Looking for More About the Latest Research?
If you would like more information about the latest areas of research in prostate cancer, explore these related items that take you outside of this guide:
To find clinical trials specific to your diagnosis, talk with your doctor or search online clinical trial databases .
Visit the Cancer.Net Blog to review research in prostate cancer, including the 2022 Genitourinary Cancers Symposium and 2021 ASCO Annual Meeting , and to listen to podcasts with experts in the field discussing recent research.
Visit the website of Conquer Cancer, the ASCO Foundation , to find out how to help support cancer research. Please note that this link takes you to a different ASCO website.
The next section in this guide is Coping with Treatment . It offers some guidance on how to cope with the physical, emotional, social, and financial changes that cancer and its treatment can bring. Use the menu to choose a different section to read in this guide.
Prostate Cancer Guide
Cancer.Net Guide Prostate Cancer
- Introduction
- Medical Illustrations
- Risk Factors and Prevention
- Symptoms and Signs
- Stages and Grades
- Types of Treatment
- About Clinical Trials
- Latest Research
- Coping with Treatment
- Follow-Up Care
- Questions to Ask the Health Care Team
- Additional Resources
View All Pages
Timely. Trusted. Compassionate.
Comprehensive information for people with cancer, families, and caregivers, from the American Society of Clinical Oncology (ASCO), the voice of the world's oncology professionals.
Find a Cancer Doctor
Prostate cancer: Newly-developed inhibitor shows massive potential
More than 65,000 men fall ill with prostate cancer each year in Germany. Twelve thousand of them develop a treatment-resistant form which eventually ends in death. Now, a team of researchers from the Medical Faculty at the University of Freiburg has developed an active substance that might in future represent a new treatment option. This substance, known as KMI169, targets an enzyme that plays an important role in the development of prostate cancer. The inhibitor displayed massive potential in among others cancer cells that were resistant to conventional treatments.
Researchers from the Department of Urology at the Freiburg University Medical Center as well as the Institut für Pharmazeutische Wissenschaften at the University of Freiburg published their study in Nature Communications on 2 January 2024 .
"We've had our eye on the enzyme KMT9 as a possible target in prostate cancer for a long time. The development of this specific inhibitor is now a decisive step in combating prostate cancer far more effectively," explains study head Professor Roland Schüle, Academic Director of the Department of Urology at the Freiburg University Medical Center and Dr. Eric Metzger, group leader in Schüle's department.The substance's potential use against treatment-resistant forms of cancer makes it especially valuable. "This treatment-resistance means that the classic antihormonal treatment often fails within a few months and the disease then progresses rapidly. The inhibitor we've developed offers us a highly innovative therapeutic approach here," says Schüle.
New approach also relevant to bladder cancer
Using cell cultures, the groups headed by Schüle and co-author Professor Manfred Jung, head of the Chemical Epigeneticsgroup of the Institut für Pharmazeutische Wissenschaften, have shown that the enzyme KMT9, known as a methyltransferase, is a critical factor in the development and progress of certain types of cancer such as prostate or bladder cancer. "The inhibitor fits snugly like a key in its lock and blocks the functioning of KMT9 and therefore also the growth of both prostate and bladder cancer cells," says Jung. The development of KMI169 was guided by crystal structure analysis of KMT9 and numerous other studies. "We modified the compound many times to increase its potency, selectivity and medicinal properties."
- Men's Health
- Prostate Cancer
- Breast Cancer
- Diseases and Conditions
- Prostate Health
- Lung Cancer
- Prostate cancer
- Colorectal cancer
- Breast cancer
- Malignant melanoma
- Ovarian cancer
- Stomach cancer
- Cervical cancer
- Hip dysplasia
Story Source:
Materials provided by University of Freiburg . Note: Content may be edited for style and length.
Journal Reference :
- Sheng Wang, Sebastian O. Klein, Sylvia Urban, Maximilian Staudt, Nicolas P. F. Barthes, Dominica Willmann, Johannes Bacher, Manuela Sum, Helena Bauer, Ling Peng, Georg A. Rennar, Christian Gratzke, Katrin M. Schüle, Lin Zhang, Oliver Einsle, Holger Greschik, Calum MacLeod, Christopher G. Thomson, Manfred Jung, Eric Metzger, Roland Schüle. Structure-guided design of a selective inhibitor of the methyltransferase KMT9 with cellular activity . Nature Communications , 2024; 15 (1) DOI: 10.1038/s41467-023-44243-6
Cite This Page :
Explore More
- 3D Mouth of an Ancient Jawless Fish
- Connecting Lab-Grown Brain Cells
- Device: Self-Healing Materials, Drug Delivery
- How We Perceive Bitter Taste
- Next-Generation Digital Displays
- Feeling Insulted? How to Rid Yourself of Anger
- Pregnancy Accelerates Biological Aging
- Tiny Plastic Particles Are Found Everywhere
- What's Quieter Than a Fish? A School of Them
- Do Odd Bones Belong to Gigantic Ichthyosaurs?
Trending Topics
Strange & offbeat.
Thank you for visiting nature.com. You are using a browser version with limited support for CSS. To obtain the best experience, we recommend you use a more up to date browser (or turn off compatibility mode in Internet Explorer). In the meantime, to ensure continued support, we are displaying the site without styles and JavaScript.
- View all journals
- Explore content
- About the journal
- Publish with us
- Sign up for alerts
- 14 September 2022
Prostate cancer: highlights from research
- Annette Fenner
You can also search for this author in PubMed Google Scholar
Armoured immunotherapy
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
24,99 € / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
185,98 € per year
only 3,65 € per issue
Rent or buy this article
Prices vary by article type
Prices may be subject to local taxes which are calculated during checkout
Nature 609 , S32-S33 (2022)
doi: https://doi.org/10.1038/d41586-022-02857-8
This article is part of Nature Outlook: Prostate cancer , an editorially independent supplement produced with the financial support of third parties. About this content.
Related Articles

Sponsor feature: How to get in front of prostate cancer
- Therapeutics
- Health care
- Cell biology
How to supercharge cancer-fighting cells: give them stem cell skills
News 10 APR 24
FOXO1 is a master regulator of memory programming in CAR T cells
Article 10 APR 24
The PARTNER trial of neoadjuvant olaparib in triple-negative breast cancer
Article 08 APR 24
Anti-ageing antibodies revive the immune system
News & Views 27 MAR 24

How AI is being used to accelerate clinical trials
Nature Index 13 MAR 24

Yaws could soon be eradicated — 70 years behind schedule
Outlook 11 JAN 24

Bird flu outbreak in US cows: why scientists are concerned
News Explainer 08 APR 24
Time to sound the alarm about the hidden epidemic of kidney disease
Editorial 03 APR 24
Adopt universal standards for study adaptation to boost health, education and social-science research
Correspondence 02 APR 24
Equipment Service Technician
Memphis, Tennessee
St. Jude Children's Research Hospital (St. Jude)
Supv-Environmental Services
Biomedical technician, maintenance assistant - liquid nitrogen handling, building automation systems engineer (hvac and lighting).
Sign up for the Nature Briefing newsletter — what matters in science, free to your inbox daily.
Quick links
- Explore articles by subject
- Guide to authors
- Editorial policies
Together we are beating cancer
- Cancer types
- Breast cancer
- Bowel cancer
- Lung cancer
- Prostate cancer
- Cancers in general
- Clinical trials
- Causes of cancer
- Coping with cancer
- Managing symptoms and side effects
- Mental health and cancer
- Money and travel
- Death and dying
- Cancer Chat forum
- Health Professionals
- Cancer Statistics
- Cancer Screening
- Learning and Support
- NICE suspected cancer referral guidelines
- Make a donation
- By cancer type
- Leave a legacy gift
- Donate in Memory
- Find an event
- Race for Life
- Charity runs
- Charity walks
- Search events
- Relay for Life
- Volunteer in our shops
- Help at an event
- Help us raise money
- Campaign for us
- Do your own fundraising
- Fundraising ideas
- Get a fundraising pack
- Return fundraising money
- Fundraise by cancer type
- Set up a Cancer Research UK Giving Page
- Find a shop or superstore
- Become a partner
- Cancer Research UK for Children & Young People
- Our Play Your Part campaign
- Brain tumours
- Skin cancer
- All cancer types
- By cancer topic
- New treatments
- Cancer biology
- Cancer drugs
- All cancer subjects
- All locations
- By Researcher
- Professor Duncan Baird
- Professor Fran Balkwill
- Professor Andrew Biankin
- See all researchers
- Our achievements timeline
- Our research strategy
- Involving animals in research
- Research opportunities
- For discovery researchers
- For clinical researchers
- For population researchers
- In drug discovery & development
- In early detection & diagnosis
- For students & postdocs
- Our funding schemes
- Career Development Fellowship
- Discovery Programme Awards
- Clinical Trial Award
- Biology to Prevention Award
- View all schemes and deadlines
- Applying for funding
- Start your application online
- How to make a successful applicant
- Funding committees
- Successful applicant case studies
- How we deliver research
- Our research infrastructure
- Events and conferences
- Our research partnerships
- Facts & figures about our funding
- Develop your research career
- Recently funded awards
- Manage your research grant
- Notify us of new publications
- Find a shop
- Volunteer in a shop
- Donate goods to a shop
- Our superstores
- Shop online
- Wedding favours
- Cancer Care
- Flower Shop
- Our eBay store
- Shoes and boots
- Bags and purses
- We beat cancer
- We fundraise
- We develop policy
- Our global role
- Our organisation
- Our strategy
- Our Trustees
- CEO and Executive Board
- How we spend your money
- Early careers
- Your development
Cancer News
- For Researchers
- For Supporters
- Press office
- Publications
- Update your contact preferences
- About cancer
- Get involved
- Our research
- Funding for researchers
The latest news, analysis and opinion from Cancer Research UK
- Science & Technology
- Health & Medicine
- Personal Stories
- Charity News
- Health & Medicine
Can prostate cancer screening with the PSA test help to save lives?

6 April 2024

- The UK National Screening Committee currently doesn’t recommend the PSA test for prostate cancer screening in men without symptoms
- The most recent studies show that PSA testing has little impact on reducing deaths from prostate cancers
- Using PSA testing for prostate screening leads to prostate cancers being ‘overdiagnosed’ and lead to some people undergoing unnecessary treatment
The earlier a cancer is diagnosed the better. That’s because when it isn’t too large and hasn’t spread there are generally more treatment options available.
One way in which we can help diagnose cancers earlier is through screening programmes. This is when people without cancer symptoms are tested for the disease.
Currently, there are three screening programmes in the UK , for breast, bowel and cervical cancer. These programmes help to save thousands of lives each year. England is also in the process of introducing a new targeted lung cancer screening programme for people at high risk of the disease.
But why isn’t there a screening programme for prostate cancer? Here, we take a look at some of the latest research into PSA testing to find out.
What is the PSA test?
The PSA test measures the amount of prostate specific antigen (PSA) in the blood. PSA is a protein made by both normal and cancerous prostate cells.
Its normal for all men to have some PSA in their blood. High levels of PSA can be a sign of prostate cancer, but PSA levels can also be raised because of other conditions, such as a urine infection.
It’s important to note that a prostate cancer diagnosis is not made on PSA level alone. Instead, PSA testing can be used to help a doctor to decide whether to undertake further tests, such as an MRI scan or biopsy.
Why is the PSA test not used for prostate screening?
You may have heard people suggesting the PSA test be used to screen people for prostate cancer, or you may even know someone who had the PSA test when they had no symptoms.
But currently the UK National Screening Committee (NSC) – the independent organisation that reviews the evidence for screening programmes – does not recommend PSA testing for prostate screening because the benefits do not outweigh the harms.
That is because the PSA test is not reliable or accurate enough at finding prostate cancer. This means that it can miss some cancers (false negative) or can suggest someone has cancer when they don’t (false positive). It can also lead to overdiagnosis.
The problem of overdiagnosis
Not all prostate cancers are the same. Some grow fast and spread quickly, while others grow slowly. Some of these cancers grow so slowly (or not at all) that if they went undetected they wouldn’t cause any problems and the person wouldn’t be harmed by their cancer if it was left untreated.
When these harmless cancers are found, they are said to be ‘overdiagnosed’.
Because it’s impossible to tell the potentially harmful prostate cancers from the harmless ones, men who are diagnosed with prostate cancer are usually offered treatment. So in some cases, men are unnecessarily treated for prostate cancer that would never have harmed them.
These treatments can have a serious impact on people’s lives, including the possibility of infection following a biopsy and erectile dysfunction and bladder problems following treatment.
What does the latest research show?
The CAP trial is the largest ever study into prostate cancer screening to date. It included over 400,000 men aged 50-69 spanning almost 600 GP practices across the UK.
Recently, the 15-year follow up of the study was published in the American Medical Association (JAMA) . The study found that a single invitation to a PSA blood test for men without symptoms had little impact on reducing prostate cancer deaths, reducing mortality by less than 1 man for every 1,000 invited for screening.
The results of the trial also showed that an estimated 1 in 6 cancers found by the single PSA screening were overdiagnosed which may have led to unnecessary and invasive treatment.
There have been improvements in diagnosing and treating the disease in the years since this trial began, including the introduction of MRI before biopsy . These changes may help to prevent some harms associated with PSA testing, but more research is needed on ways to find aggressive cancers that need to be treated.
Black men and family history
We know that PSA testing in men without symptoms is not suitable for the general population. But what about people who may be at a higher risk of prostate cancer, such as Black men or men who have a family history of prostate cancer? Well first we would have to know if these men are at a higher risk of a harmful prostate cancer or at a higher risk of overdiagnosis.
Some research has shown that Black men have naturally higher levels of PSA than White men . This means using the PSA test in Black men may lead to higher levels of overdiagnosis. And while proportionally more Black men are dying from prostate cancer in England, once diagnosed with prostate cancer Black men have the same chance as White men of dying from the disease which could suggest that Black men are not at a higher risk of harmful prostate cancer.
Overall, current research has not shown that PSA testing in those with no symptoms saves lives in the general population, but there is not enough research on PSA testing in Black men and those with a family history of prostate cancer. Future research should look into this and other tests that can more accurately diagnose prostate cancers that needs treatment.
What are Cancer Research UK doing?
To try and save more lives from the disease, Cancer Research UK is funding more trials and research into prostate cancer. This includes speeding up diagnosis for men with symptoms so they can start treatment sooner, and new blood, urine or genetic tests to improve early diagnosis.
Other research, such as the STAMPEDE trial , is aiming to find the best treatment for men with advanced prostate cancer to further improve survival and quality of life. The charity has spent £21 million on prostate cancer research over the last two years to improve outcomes for patients.
What should men look out for?
Cancer Research UK raises awareness of the importance of listening to your body and getting to know what’s normal for you. If you notice a change that doesn’t go away or is new, then speak to your doctor.
If a man has urinary symptoms, such as such as difficulty peeing or needing to pee more often – most of which are caused by a non-cancerous enlargement of the prostate – he can contact his GP for advice, and this may include a PSA test.
My GP told me the PSA test is largely a waste of time (I’m in my 70s). Clearly more research is needed to develop a test which is at least reasonably reliable.
Tell us what you think
Leave a reply cancel reply.
Your email address will not be published. Required fields are marked *
Save my name, email, and website in this browser for the next time I comment.
Read our comment policy .
Highlighted content

That Cancer Conversation podcast – Longer, better lives: Ep.2 Why did a doctor have to wait for her cancer treatment?

What's it like to be diagnosed with cancer as a teenager?

£1.7 million for the world’s first vaccine to prevent lung cancer

Can vaping cause changes in our cells?

A message to MPs: Don’t miss the opportunity to make smoking history
From lemonade stand to grand challenge: changing how we treat cancer in children and young people

Cancer Grand Challenges gives a record £100m to five cancer research teams
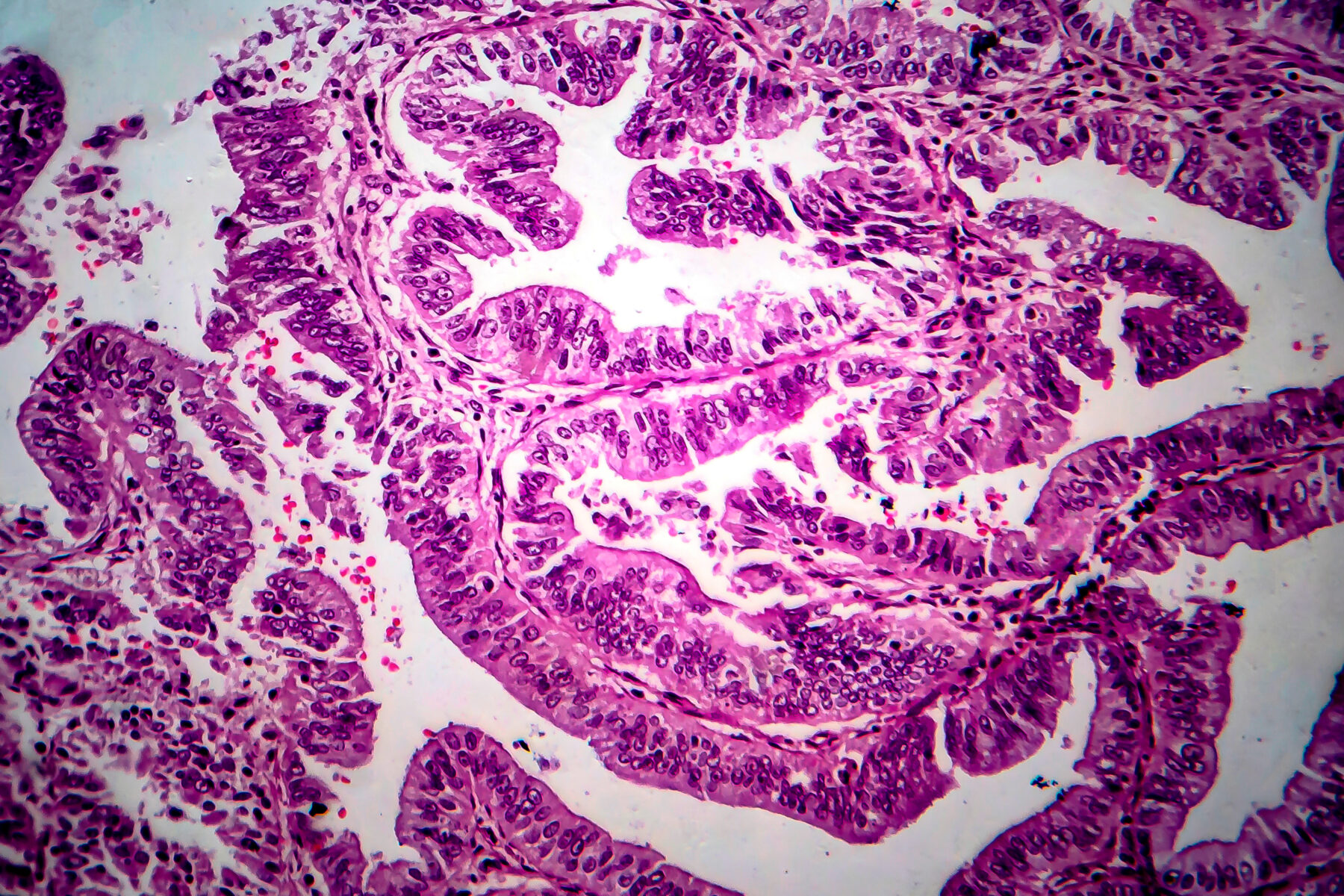
New treatment option made available for advanced womb cancers

Cancer deaths plummet in middle-aged people
Related topics.
- Early detection
- Cancer in the news

IMAGES
VIDEO
COMMENTS
NCI funds and oversees both early- and late-phase clinical trials to develop new treatments and improve patient care. Trials are available for prostate cancer prevention, screening, and treatment. Prostate Cancer Research Results. The following are some of our latest news articles on prostate cancer research:
Current research aims to improve prostate cancer detection, management and outcomes, including understanding the fundamental biology at all stages of the disease. ... In addition, more than 1.2 ...
Currently used prostate cancer treatments have serious adverse effects; therefore, new research is focusing on alternative treatment options such as the use of genetic biomarkers for targeted gene therapy, nanotechnology for controlled targeted treatment, and further exploring medicinal plants for new anticancer agents.
Between 1999 and 2009 in the United Kingdom, 82,429 men between 50 and 69 years of age received a prostate-specific antigen (PSA) test. Localized prostate cancer was diagnosed in 2664 men. Of these...
Prostate cancer is a type of malignancy that arises in the prostate gland. Prostate cancer tends to develop in older men, aged 50 and over. ... Latest Research and Reviews. ... Research Open ...
The management of prostate cancer continues to evolve rapidly, with substantial advances being made in understanding the genomic landscape and biology underpinning both primary and metastatic prostate cancer. Similarly, the emergence of more sensitive imaging methods has improved diagnostic and staging accuracy and refined surveillance strategies. These advances have introduced personalised ...
Prostate cancer is a substantial health concern worldwide and, currently, the most commonly diagnosed malignancy in men in the USA 1.Surgery and/or radiation therapy is available for localized ...
When PARP is blocked by treatment, the cancer cells will eventually die. Other PARP inhibitors, including olaparib and rucaparib, are already approved for advanced prostate cancer in BRCA-positive men. During research leading to this latest approval, 399 men with mCRPC were randomly divided into two groups.
Prostate cancer treatment took a major step forward today as the U.S. Food and Drug Administration approved a new therapy that zeros in on cancer cells to destroy them. The treatment, called 177 Lu-PSMA-617, uses a molecule that selectively seeks out and attaches to a specific protein on the cancer cell surface called PSMA (prostate-specific membrane antigen).
ExoDx Prostate (IntelliScore), or EPI, a test that looks at levels of 3 biomarkers in a urine sample to help determine a man's risk of having aggressive (high-grade) prostate cancer. SelectMDx, which looks at the levels of certain forms of RNA (linked to certain cancer-related genes) in the urine. This test can be used along with other ...
Learn the symptoms of prostate cancer. Read current medical research on new treatment options, vaccines, surgery and prevention.
Thursday, June 3, 2021. Michael Rosenblum received an experimental new prostate cancer treatment after the disease spread to his bones. Since then, he has been symptom-free. Update: On March 23, 2022, the US Food and Drug Administration approved 177 Lu-PSMA-617 for prostate cancer that has spread and grown resistant to other drugs.
Prostate cancer is the most common cancer in men in 112 countries, and accounts for 15% of cancers. In this Commission, we report projections of prostate cancer cases in 2040 on the basis of data for demographic changes worldwide and rising life expectancy. Our findings suggest that the number of new cases annually will rise from 1·4 million in 2020 to 2·9 million by 2040.
One of these new therapies — a sort of smart bomb targeted at malignant cells — is now generating promising data for men with the most aggressive prostate cancer. In early June, investigators reported results from a phase 3 clinical trial showing that among men who received the experimental treatment, there was nearly a 40% reduction in ...
Sep. 21, 2022 —. Nov. 2, 2021 —. Nov. 27, 2019 —. Aug. 27, 2019 —. Investigators have identified two promising new treatment options for men with recurrent prostate cancer -- both of which ...
A globe-spanning scientific team has compiled the most comprehensive list of genetic variants associated with prostate cancer risk -- 451 in all -- through a whole-genome analysis that ranks as ...
The study was funded by Prostate Cancer UK, Cancer Research UK, the Swiss Card Onco grant organisation, the Prostate Cancer Foundation, AstraZeneca, Wellcome and the NIHR Biomedical Research ...
Prostate cancer is the most common solid cancer in men worldwide. The number of estimated new prostate cancers in the US in 2022 was 268,490. The number of estimated deaths due to prostate cancer is 34,500 . Surgery and radiotherapy are the mainstay of treatment in localized disease.
Prostate cancer is already a major cause of morbidity and mortality worldwide and the prostate cancer commission projects that the number of new cases will double between 2020-2040. The commission analysis both immediate and long-term interventions to mitigate the current and projected future global impact of prostate cancer.
Prostate-specific membrane antigen positron emission tomography (PSMA-PET) is a novel imaging method that was shown to be more accurate than conventional imaging in prostate cancer staging, and ...
New, advanced treatments for prostate cancer provide patients with a multitude of options. Curtiland Deville, M.D., medical director of the Johns Hopkins Proton Therapy Center at Sibley Memorial Hospital and co-director of the Johns Hopkins Kimmel Cancer Center Prostate Multidisciplinary Clinic at Sibley Memorial Hospital, says that the team works with each patient to identify the most ...
Recent research from the University of Eastern Finland sheds light on the significance of the glucocorticoid receptor in drug-resistant prostate cancer, showing that the development of drug ...
Visit the Cancer.Net Blog to review research in prostate cancer, including the 2022 Genitourinary Cancers Symposium and 2021 ASCO Annual Meeting, and to listen to podcasts with experts in the field discussing recent research. Visit the website of Conquer Cancer, the ASCO Foundation, to find out how to help support cancer research.
Jan. 5, 2021 — New research has identified a new mechanism in which prostate cancer cells can 'switch' character and become resistant to therapy. These findings are an important development in ...
During the research, Dr. Kaplan's team measured changes in the International Prostate Symptom Score (IPSS), which ranges from 0 to 35 and classifies BPH as either mild, moderate, or severe. According to results from the first clinical trial, called the PINNACLE study and limited to men with prostates ranging from 20 to 80 grams in size ...
Part of Nature Outlook: Prostate cancer. The researchers developed CAR-T cells that target prostate-specific membrane antigen (PSMA) — a protein that is highly expressed in prostate cancer cells ...
Prostate cancer is the most common form of cancer that affects British men, with about 52,000 cases annually, according to Cancer Research UK senior science engagement manager Rupal Mistry.
What are Cancer Research UK doing? To try and save more lives from the disease, Cancer Research UK is funding more trials and research into prostate cancer. This includes speeding up diagnosis for men with symptoms so they can start treatment sooner, and new blood, urine or genetic tests to improve early diagnosis.
Scientists at the London Institute of Cancer Research predict there will be 2.9 million annual cases of prostate cancer around the world by 2040, more than twice as many as the 1.4 million cases ...